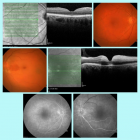
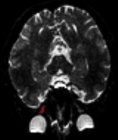
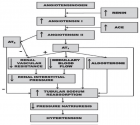
Annals of Clinical Endocrinology and Metabolism | ISSN: 2640-284X

License:
Copyright © 2016 - 2024 |  Open Access by Annals of Clinical Endocrinology and Metabolism is licensed under a Creative Commons Attribution 4.0 International License. Based on a work at Heighten Science Publications Inc.
Open Access by Annals of Clinical Endocrinology and Metabolism is licensed under a Creative Commons Attribution 4.0 International License. Based on a work at Heighten Science Publications Inc.
Explore Journal
Why with Us
Annals of Clinical Endocrinology and Metabolism has a rich history and track-record of delivering simpler and updated access to the research needs in
Read MoreFor Editors
Annals of Clinical Endocrinology and Metabolism trail a critical evaluation prior to selection of Editorial Board members. In order to streamline our
Read MoreAbout Journal
Annals of Clinical Endocrinology and Metabolism is a distinct division of Health Care Sector, which takes care of physical fitness and injuries associ
Read MoreMembership
There is a precept- "two heads result in more fruitful thing than one head alone". Annals of Clinical Endocrinology and Metabolism unique concept of m
Read MoreExplore Universities
Editorial Board
Most Viewed Keywords
- Orgasm
- Linear No-Threshold (LNT) model
- Thrombocytopenic purpura-haemolytic uremic syndrome
- SARS-CoV-2
- Acyclovir
- Emphysematous pyelonephritis
- Strophanthus hispidus
- Dysplasia diagnosis
- Rat knee
- Bone marrow donation
- Emotions
- Children
- Hypovolemic shock
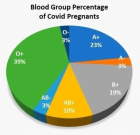
- COVID-19
- Fregoli
- Beta-adrenergic blocking
- Intraoperative hemofiltration
- Aortic valve stenosis
- Medical marijuana
- Thyroid
Latest Articles

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."