More Information
Submitted: March 26, 2026 | Approved: April 10, 2026 | Published: April 13, 2026
How to cite this article: Vanichakulthada N, Pitchaiprasert S, Jaroenying R, Hantragool S, Samhugkanee C. Sustainable Weight Loss and Body Composition Improvement through Intensive Lifestyle Modification with Meal Replacement: A 12-Case Series with International Perspectives. Ann Clin Endocrinol Metabol. 2026; 10(1): 017-025. Available from:
https://dx.doi.org/10.29328/journal.acem.1001034.
DOI: 10.29328/journal.acem.1001034
Copyright License: © 2026 Vanichakulthada N, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Obesity; Intensive lifestyle modification; Meal replacement; Weight loss; Case series; Body composition; Global health; Asia-Pacific
Sustainable Weight Loss and Body Composition Improvement through Intensive Lifestyle Modification with Meal Replacement: A 12-Case Series with International Perspectives
Nawinda Vanichakulthada1*, Sunaree Pitchaiprasert2, Rasintra Jaroenying3, Sumalee Hantragool4 and Chutima Samhugkanee5
1College of Medicine and Public Health, Ubon Ratchathani University, Ubon Ratchathani, Thailand
2Department of Obstetrics and Gynecology, Surin Hospital, Surin, Thailand
3Department of Pediatrics, Phramongkutklao Hospital, Thailand
4Department of Pediatrics, Piyavate Hospital, Thailand
5Independent Researcher, Thailand
*Address for Correspondence: Nawinda Vanichakulthada, College of Medicine and Public Health, Ubon Ratchathani University, Ubon Ratchathani, Thailand, Email: [email protected]
Background: Obesity is one of the most pressing global health challenges, with over one billion people now living with the condition worldwide. In Southeast Asia, nearly 40% of the population is projected to be overweight or obese by 2035, with Thailand reporting prevalence exceeding 42%. Intensive Lifestyle Modification (ILM) combined with Meal Replacement (MR) has emerged as a promising strategy for achieving sustainable weight loss, supported by landmark trials including the Diabetes Prevention Program, Look AHEAD, and DiRECT.
Objective: To illustrate the clinical effectiveness of ILM combined with MR through detailed case presentations documenting anthropometric outcomes at baseline, 8 weeks, and 52 weeks, contextualized within international obesity management guidelines and comparative research across diverse populations. We hypothesized that selected participants undergoing the ILM+MR intervention would demonstrate clinically significant weight loss (≥5% of initial body weight) sustained through 52 weeks, with concurrent improvements in waist circumference indicating reduced central adiposity.
Methods: Twelve participants (9 female, 3 male) from the ILM+MR arm of a previously published retrospective cohort study (n = 702) were selected based on complete longitudinal data and representativeness. The intervention comprised structured nutritional counseling, soy-based MR (220 kcal per serving, twice daily for 8 weeks), behavioral modification with group therapy, and physical activity guidance aligned with the 2013 AHA/ACC/TOS guidelines. Body weight and waist circumference were measured at baseline, 8 weeks, and 52 weeks.
Results: Mean total weight loss was 38.2 kg (35.3% of initial body weight), with mean waist circumference reduction of 13.0 inches (33.0 cm) over 52 weeks. All 12 participants achieved clinically significant weight loss (>5%), substantially exceeding the 5–10% threshold recommended by international guidelines. Weight loss occurred in two phases: a rapid intensive phase (mean 14.0 kg during weeks 0–8) followed by continued loss during maintenance (mean 24.2 kg during weeks 8–52). All participants completed the 52-week follow-up.
Conclusion: This case series demonstrates the potential for substantial, sustained weight loss through ILM+MR intervention, supporting findings from the larger Thai cohort study and international research including the DiRECT trial and systematic meta-analyses. These results reaffirm the foundational role of comprehensive lifestyle programs and highlight their relevance as scalable, culturally adaptable interventions for obesity management across diverse populations.
The global obesity crisis
Obesity has emerged as one of the most significant public health challenges of the modern era. The World Health Organization reports that more than one billion people worldwide now live with obesity, and global prevalence has nearly tripled since 1975 [1]. This epidemic extends across all geographic regions and socioeconomic strata, with the economic burden estimated at $2 trillion annually in direct healthcare costs and indirect productivity losses [2,3]. The urgency of effective intervention has been amplified by the recognition that obesity is a complex chronic disease requiring lifelong, multifaceted management rather than episodic treatment [4].
Regional perspectives: The Asia-Pacific obesity epidemic
The Asia-Pacific region has experienced particularly rapid increases in obesity prevalence, driven by urbanization, nutrition transition toward calorie-dense diets, and decreased physical activity. In Southeast Asia, nearly 40% of the population is projected to be overweight or obese by 2035, with over 330,000 annual deaths attributed to obesity-related complications across ASEAN countries [5,6]. In Thailand, national health surveys indicate that over 42% of the population is now classified as overweight or obese, representing one of the highest prevalence rates in the region [7]. The direct and indirect costs of overweight and obesity in Southeast Asia were estimated at $11.3 billion in 2018, representing a substantial burden on healthcare systems striving toward universal health coverage [5].
These trends are particularly concerning given the higher metabolic risk associated with central adiposity in Asian populations at lower Body Mass Index (BMI) thresholds compared to Western counterparts. The WHO Expert Consultation recommended Asian-specific BMI cutoffs (≥25 kg/m² for obesity) based on evidence that cardiometabolic risk increases at lower adiposity levels in these populations [8]. A recent consensus statement on obesity management in South and Southeast Asia further emphasized the need for ethnicity-specific approaches, noting that 5% - 15% weight loss over 6 months represents a safe and realistic goal for most patients, while greater reductions may be needed for specific clinical endpoints such as diabetes remission [9].
Evidence-based lifestyle interventions: The foundation of obesity management
Intensive lifestyle modification incorporating dietary changes, physical activity, and behavioral interventions remains the cornerstone of obesity management, as established by the 2013 AHA/ACC/TOS guidelines [10]. The evidence base for this approach is anchored in several landmark trials. The Diabetes Prevention Program (DPP) demonstrated that intensive lifestyle intervention achieving 7% weight loss reduced diabetes incidence by 58%, with benefits persisting over 10 years [11,12]. The Look AHEAD study showed sustained improvements in weight, cardiovascular risk factors, and quality of life over 8 years [13]. However, adherence remains a persistent challenge, with attrition rates typically ranging from 30–60% across diverse cultural and socioeconomic contexts [14,15].
The incorporation of Meal Replacements (MRs) into lifestyle programs has shown promise in addressing the adherence gap. The meta-analysis by Heymsfield, et al. pooling data from six randomized controlled trials demonstrated that MR strategies produced significantly greater weight loss compared to conventional diets [16]. The DiRECT trial by Lean, et al. provided landmark evidence that intensive weight management incorporating total diet replacement achieved 46% type 2 diabetes remission at 12 months, with weight loss strongly predicting remission probability [17]. These findings have transformed clinical perspectives on the potential for lifestyle intervention to induce disease remission.
Rationale for the current case series
Our previously published retrospective cohort study (n = 702) demonstrated that ILM combined with MR achieved superior outcomes compared to ILM alone and a control group over 52 weeks, with mean weight loss of 14.43 kg, BMI reduction of 5.54, and waist circumference decrease of 7.41 inches in the ILM+MR group (p < 0.001), alongside a 7.54% reduction in fat mass and 2.33% increase in muscle mass [18]. The present case series presents 12 individual participants selected for detailed analysis to illustrate the clinical effectiveness and real-world applicability of the intervention, contextualized within the international research landscape. We hypothesized that selected participants undergoing the ILM+MR intervention would achieve clinically significant weight loss (≥5% of initial body weight) sustained through 52 weeks, with concurrent reductions in waist circumference reflecting improved central adiposity, at magnitudes exceeding those reported in major international lifestyle intervention trials.
Study design and participant selection
This case series comprises 12 participants selected from the ILM+MR intervention group of our retrospective cohort study conducted between 2022 and 2023 at clinical sites in Thailand [18]. Participants were selected based on the availability of complete longitudinal data at all three assessment time points (baseline, 8 weeks, and 52 weeks) and representativeness of the broader cohort demographics. All participants provided written informed consent for publication of their anonymized clinical data.
Eligibility criteria
Participants were aged 18 - 80 years with obesity defined as BMI ≥25 kg/m² (using Asian-specific cutoffs per WHO Expert Consultation [8]) or central obesity with waist circumference >90 cm for men and >80 cm for women. Exclusion criteria included pregnancy, breastfeeding, preexisting chronic diseases requiring treatment affecting weight, and concurrent participation in other weight loss programs. The study was approved by The Ubon Ratchathani University Ethics Committee for Human Research (approval number: UBU-REC-103/2567) and conducted in accordance with the Declaration of Helsinki.
Case selection criteria for detailed presentation
From the 230 participants in the ILM+MR arm of the parent cohort study, 12 cases were selected for detailed presentation in this case series based on the following inclusion criteria: (1) availability of complete anthropometric data (body weight and waist circumference) at all three assessment time points (baseline, week 8, and week 52); (2) achievement of clinically significant weight loss (≥5% of initial body weight) at 52 weeks, consistent with international guideline thresholds [10]; (3) representation of the demographic diversity observed in the broader cohort, including both sexes, a range of baseline BMI categories (from overweight to Class III obesity), and a range of ages; and (4) demonstration of sustained weight loss trajectory through the 52-week follow-up without evidence of significant weight regain. Cases were excluded from detailed presentation if anthropometric data were incomplete at any time point, if follow-up was discontinued before 52 weeks, or if weight regain exceeded the weight lost during the intensive phase. The 12 selected cases represent participants with exceptional outcomes and are not intended to represent the full distribution of treatment responses in the parent cohort.
Intervention protocol
The ILM+MR program was designed following principles established by international guidelines and evidence from major lifestyle intervention trials, as previously described in detail [18]. The comprehensive, multi-component intervention consisted of the following:
Dietary component: A soy-based meal replacement (220 kcal per serving, containing 15 g protein, 30 g carbohydrates, 3 g fat, and complete micronutrients) was consumed twice daily, replacing breakfast and dinner during the initial 8-week intensive phase. Structured nutritional counseling emphasized high-fiber, low-glycemic-index foods for the remaining daily meal. Energy restriction targeted 1,200-1,500 kcal/day for women and 1,500-1,800 kcal/day for men, consistent with AHA/ACC/TOS recommendations [10].
Behavioral modification component: Self-monitoring through daily food and activity logs was implemented, following evidence from the STOP Regain trial [19]. Goal setting using SMART criteria, motivational group therapy sessions held bi-weekly during the intensive phase, and relapse prevention training incorporating strategies from the National Weight Control Registry research were included [20].
Physical activity component: Physical activity was gradually progressed from low-intensity walking (10 minutes post-breakfast, 5 minutes post-lunch and dinner) to moderate activity, with self-stretching exercises hourly during waking hours. The target progressed toward 150-250 minutes of moderate-intensity physical activity per week, consistent with American College of Sports Medicine recommendations [21].
Supportive components: Stress management education, sleep hygiene optimization targeting 7-9 hours nightly [22], and regular follow-up contacts through phone calls and online consultations during the maintenance phase were integral components.
Outcome measures
The primary outcomes were body weight (kg) measured using calibrated digital scales (Omron body fat meter, model HBF-375 or HBF-702T) with participants in light clothing without shoes, and waist circumference (inches) measured at the midpoint between the lowest rib and iliac crest using standardized protocols. Secondary outcomes included percentage weight loss calculated as [(baseline weight − final weight) / baseline weight] × 100. Clinically significant weight loss was defined as ≥5% of initial body weight, per international guideline recommendations [10]. Assessments were performed at baseline (week 0), post-intensive phase (week 8), and long-term follow-up (week 52).
Comparison with international benchmarks
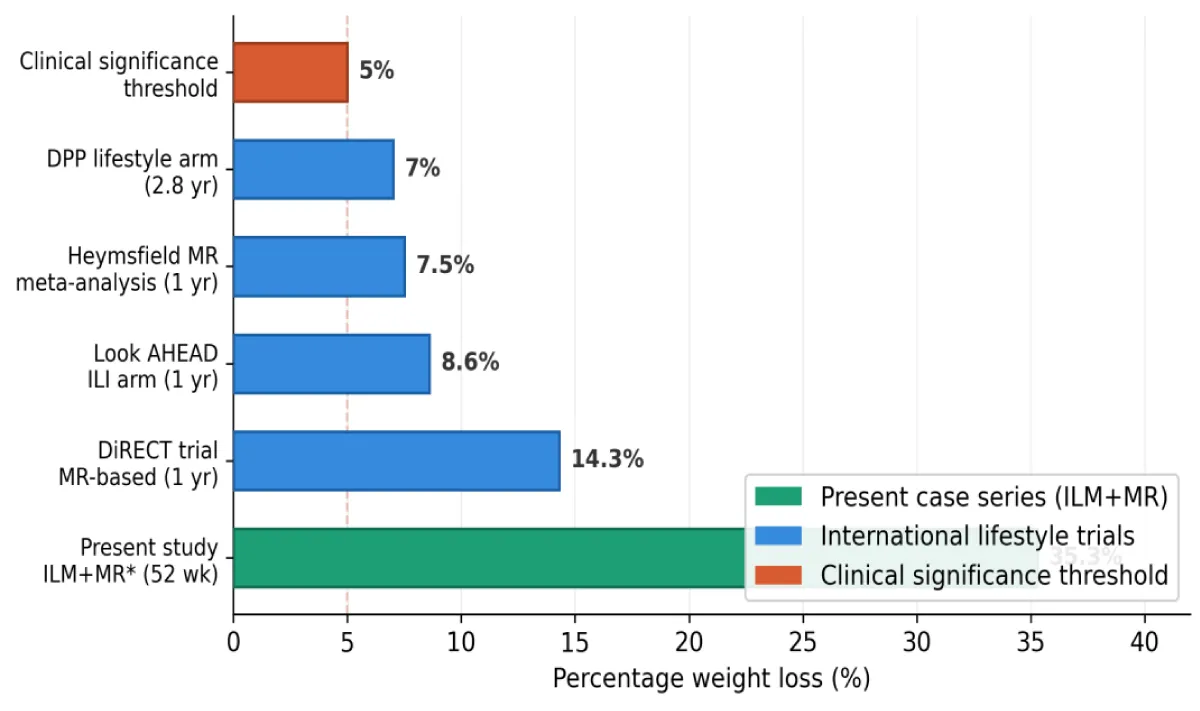
To contextualize the outcomes, results were compared against benchmarks from major international lifestyle intervention studies (Table 1 and Figure 1).
| Table 1: International benchmarks for weight loss outcomes from lifestyle interventions | |||
| Study / Intervention | Weight Loss | Duration | Reference |
| Look AHEAD (ILI arm) | 8.6% | 1 year | Look AHEAD, 2014 [13] |
| DiRECT trial (MR-based) | 10.0 kg | 1 year | Lean, et al. 2018 [17] |
| DPP lifestyle arm | 7.0% | 2.8 years | Knowler, et al. 2002 [11] |
| Heymsfield MR meta-analysis | 7–8% | 1 year | Heymsfield, et al. 2003 [16] |
| Clinical significance threshold | ≥5% | Any | AHA/ACC/TOS, 2013 [10] |
| Present case series (ILM+MR) | 35.3% | 52 weeks | Selected exceptional cases |
| ILI = Intensive Lifestyle Intervention; MR = Meal Replacement; DPP = Diabetes Prevention Program | |||
Figure 1: Comparison of percentage weight loss across international lifestyle intervention benchmarksand the present case series. DPP = Diabetes Prevention Program; DiRECT = Diabetes Remission Clinical Trial; MR = meal replacement. *Selected exceptional cases; not representative of intention-to-treat populations.
Participant characteristics
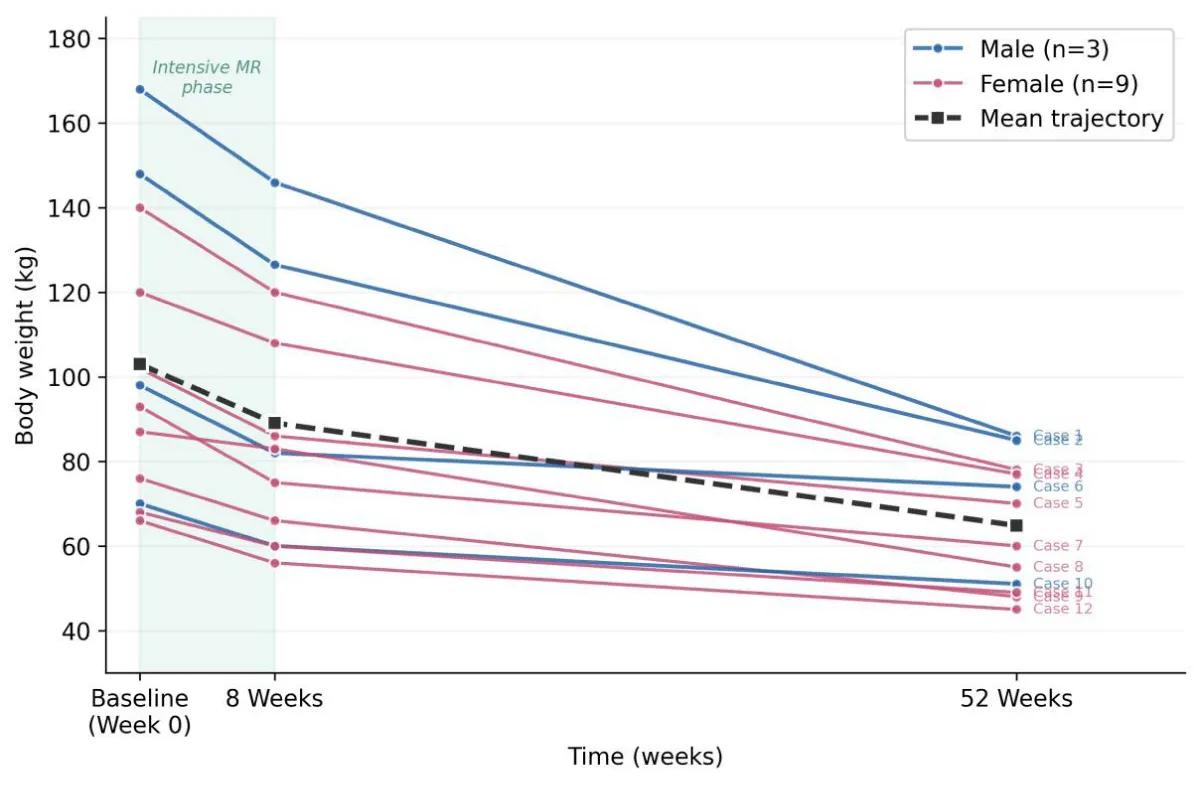
The case series includes 12 participants (9 female, 3 male), reflecting the female predominance (78.3%) observed in the larger cohort study [18]. This demographic pattern aligns with international studies showing higher treatment-seeking rates among women with obesity. Baseline weights ranged from 66 kg to 168 kg (mean 103.0 kg), and waist circumferences ranged from 36 to 58 inches (mean 44.7 inches). All participants completed the 52-week follow-up, representing complete retention. Individual participant outcomes are summarized in Table 2, with weight loss trajectories illustrated in Figure 2.
Figure 2: Individual weight loss trajectories for 12 participants at baseline, 8 weeks, and 52 weeks. Each line represents one participant. Blue lines indicate male participants; pink lines indicate female participants. The dashed line shows the mean trajectory. The shaded region indicates the 8-week intensive meal replacement phase.
| Table 2: Individual case outcomes at baseline, 8 weeks, and 52 weeks. | |||||||||
| Case | Sex | Baseline Wt (kg) | Baseline WC (in) | 8-Week Wt (kg) | 8-Week WC (in) | 52-Week Wt (kg) | 52-Week WC (in) | Total Loss (kg) | % Weight Loss |
| 1 | M | 168 | 56.5 | 146 | 50 | 86 | 37 | 82 | 48.8% |
| 2 | M | 148 | 58 | 126.5 | 50 | 85 | 38 | 63 | 42.6% |
| 3 | F | 140 | 48 | 120 | 40 | 78 | 32 | 62 | 44.3% |
| 4 | F | 120 | 46 | 108 | 41 | 77 | 34 | 43 | 35.8% |
| 5 | F | 102 | 48 | 86 | 42 | 70 | 39 | 32 | 31.4% |
| 6 | M | 98 | 40 | 82 | 36 | 74 | 33 | 24 | 24.5% |
| 7 | F | 93 | 45 | 75 | 40 | 60 | 32 | 33 | 35.5% |
| 8 | F | 87 | 44 | 83 | 43 | 55 | 28 | 32 | 36.8% |
| 9 | F | 76 | 37.5 | 66 | 32.5 | 48 | 26 | 28 | 36.8% |
| 10 | M | 70 | 38 | 60 | 32 | 51 | 28 | 19 | 27.1% |
| 11 | F | 68 | 36 | 60 | 33 | 49 | 27 | 19 | 27.9% |
| 12 | F | 66 | 39 | 56 | 30 | 45 | 26 | 21 | 31.8% |
| Mean | — | 103.0 | 44.7 | 89.0 | 39.1 | 64.8 | 31.7 | 38.2 | 35.3% |
| Wt = Weight; WC = Waist Circumference; in = Inches; M = Male; F = Female | |||||||||
Weight loss outcomes
All 12 participants achieved substantial weight loss exceeding the clinically significant threshold of 5% recommended by international guidelines [10] (Table 2). Total weight loss ranged from 19 kg (Cases 10 and 11) to 82 kg (Case 1), with percentage weight loss ranging from 24.5% to 48.8%. The mean total weight loss was 38.2 kg, representing 35.3% of initial body weight. These outcomes substantially exceed those reported in landmark lifestyle intervention trials (Table 1; Figure 1): the Look AHEAD intensive arm reported 8.6% at 1 year [13], the DiRECT trial achieved approximately 10 kg at 1 year [17], and the DPP lifestyle arm achieved 7.0% at a mean follow-up of 2.8 years [11]. However, these comparisons must be interpreted cautiously, as the present cases were selected for favorable outcomes and do not represent intention-to-treat populations.
Waist circumference reduction and cardiometabolic implications
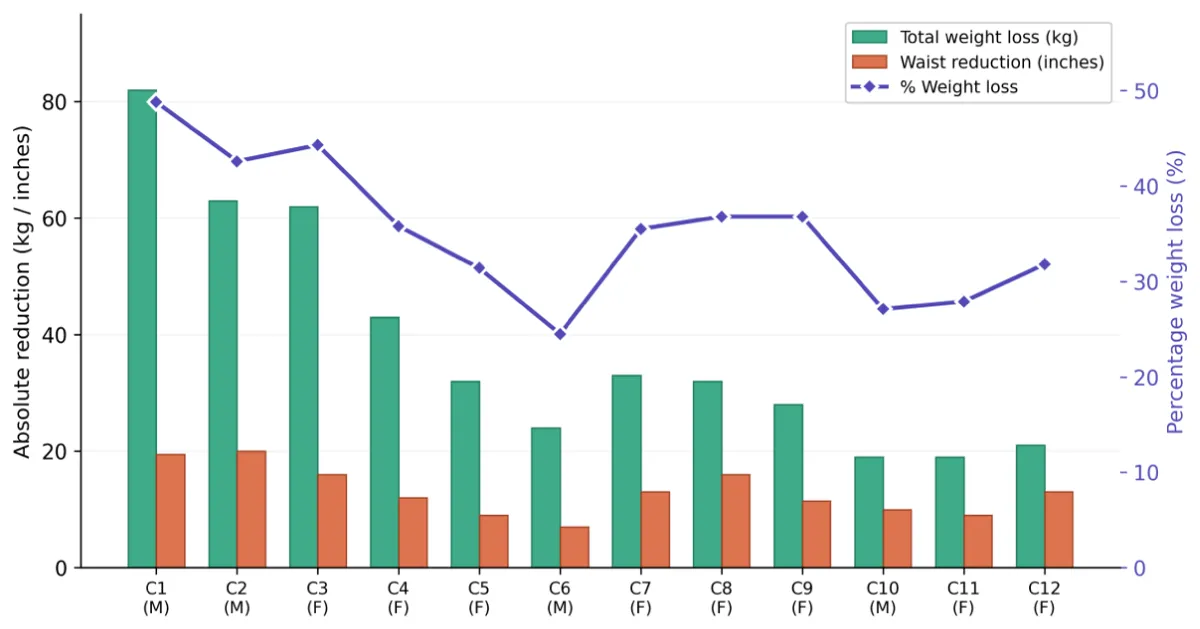
Mean waist circumference reduction was 13.0 inches (33.0 cm) over 52 weeks, ranging from 9 inches (Cases 5 and 9) to 20 inches (Case 2). The combined outcomes of weight loss, waist circumference reduction, and percentage weight loss for each individual case are presented in Figure 3. This substantial reduction in central adiposity has important implications for cardiometabolic risk, as waist circumference is strongly correlated with visceral adipose tissue and serves as an independent predictor of cardiovascular disease, type 2 diabetes, and all-cause mortality [23,24]. According to the International Diabetes Federation criteria, the threshold waist circumference values of 90 cm for Asian men and 80 cm for Asian women mark elevated cardiometabolic risk. The reductions observed moved participants substantially below these thresholds, suggesting meaningful improvement in metabolic risk profiles. This is particularly relevant in Asian populations, where the South and Southeast Asian consensus recommends ethnicity-specific approaches to central obesity assessment [9].
Figure 3: Total weight loss (kg, green bars), waist circumference reduction (inches, orange bars), and percentage weight loss (purple line, right axis) for each participant at 52 weeks. Cases are ordered by baseline body weight (heaviest to lightest). M = Male; F = Female.
Temporal patterns of weight loss
Weight loss occurred in two distinct phases. During the 8-week intensive meal replacement phase, mean weight loss was 14.0 kg (13.6% of initial body weight), representing 36.6% of total weight loss. This rapid initial loss is consistent with findings by Nackers, et al. [25] demonstrating that faster initial weight loss is associated with greater long-term success. During the maintenance phase (weeks 8–52), an additional 24.2 kg (23.5% of initial body weight) was lost, representing 63.4% of total weight loss. The continued weight loss trajectory during maintenance is notable, as many studies report plateau or regain during this period, and suggests successful internalization of lifestyle behaviors (Figure 2).
Selected case presentations
Case 1 (severe obesity with exceptional response): A male participant presented with Class III obesity, initial weight 168 kg and waist circumference 56.5 inches (143.5 cm) (Table 2). During the intensive phase, he lost 22 kg (13.1%), reducing to 146 kg by week 8. Continued lifestyle modification resulted in an additional 60 kg loss, reaching 86 kg at 52 weeks (total: 82 kg, 48.8%). This magnitude approaches outcomes reported following bariatric surgery; Buchwald, et al. [26] reported mean excess weight loss of 61.2% following Roux-en-Y gastric bypass, while the STAMPEDE trial reported 23% mean total weight loss at 5 years post-surgery [27]. Case 1 achieved approximately 70% excess weight loss through lifestyle intervention alone.
Case 3 (sustained long-term success): A female participant with initial weight 140 kg and 48-inch waist circumference demonstrated progressive weight loss: 120 kg at 8 weeks, continuing to 78 kg at 52 weeks (total: 62 kg, 44.3%). The DiRECT trial reported that 86% of participants achieving ≥15 kg weight loss maintained type 2 diabetes remission at 12 months [17]; this participant’s 62 kg loss substantially exceeds that threshold.
Case 9 (effectiveness across the BMI spectrum): A female participant with lower baseline BMI (initial weight 76 kg, waist circumference 37.5 inches) achieved a total loss of 28 kg (36.8%) and waist circumference reduction of 11.5 inches. This case demonstrates that the ILM+MR intervention produces meaningful benefits across the obesity spectrum, which is particularly relevant given that metabolic risk increases at lower BMI thresholds in Asian populations [8,9].
Case 7 (rapid initial response predicting long-term success): A female participant (initial weight 93 kg, waist circumference 45 inches) demonstrated rapid initial response with 18 kg loss by week 8, followed by continued loss to 60 kg at 52 weeks (total: 33 kg, 35.5%). The rapid initial loss of 2.25 kg/week during the intensive phase aligns with research by Nackers, et al. [25] showing that faster initial weight loss predicts greater long-term success.
Summary of findings
This case series demonstrates exceptional outcomes with the ILM+MR intervention, with mean weight loss of 38.2 kg (35.3%) and mean waist circumference reduction of 13.0 inches over 52 weeks. These results substantially exceed both the 14.43 kg mean loss reported in the ILM+MR arm of our larger cohort study (n = 230) [18] and outcomes from major international clinical trials (Table 1; Figure 1). The 100% retention rate at 52 weeks compares favorably with typical attrition rates of 30% - 60% [14,15].
Contextualization within international research
Several factors warrant consideration when interpreting these results. First, these 12 participants were selected for favorable outcomes and do not represent the full distribution of responses; the mean 35.3% weight loss should be understood as demonstrating the potential under optimal conditions rather than typical outcomes. Second, the multi-component protocol incorporates elements from multiple evidence-based approaches—structured meal replacement (DiRECT [17]), behavioral modification (DPP [11]), and comprehensive lifestyle change (Look AHEAD [13])—and may produce synergistic effects exceeding those of individual elements studied in isolation. Third, cultural and contextual factors specific to the Thai setting, including dietary patterns emphasizing rice and vegetables, family-centered social structures supporting behavioral change, and lower baseline consumption of ultra-processed foods, may have enhanced intervention effectiveness [28].
When compared to bariatric surgical outcomes, the weight loss achieved in this case series is noteworthy. Schauer, et al. [27] reported 23% mean total weight loss at 5 years following Roux-en-Y gastric bypass in the STAMPEDE trial, compared to 35.3% in the present case series. While these comparisons must be interpreted cautiously given differences in study design and population characteristics, they suggest that intensive lifestyle intervention can, in select cases, produce results comparable to surgical approaches.
Mechanisms of sustained success
Several factors likely contributed to the exceptional and sustained outcomes observed. The structured meal replacement phase provided clear dietary guidance and reduced decision fatigue, addressing the concept of self-regulatory depletion in dietary decision-making [29]. By eliminating food choices for two meals daily, participants could focus cognitive resources on behavioral change. Group therapy sessions fostered peer support and accountability, consistent with social cognitive theory principles [30] and National Weight Control Registry findings identifying social support as a key success factor [20]. The gradual transition from meal replacement to self-selected meals during weeks 8–52 allowed skills acquisition while maintaining caloric control, aligning with recommendations from the Academy of Nutrition and Dietetics [31]. The 8-week intensive phase may have been sufficient for habit formation, as research suggests behavioral automaticity develops over approximately 66 days of consistent practice [32]. Additionally, the comprehensive physical activity protocol may have partially mitigated the metabolic adaptation (reduced energy expenditure beyond that expected from weight loss) commonly observed following weight loss [33].
Sustainability and weight maintenance
The sustained weight loss observed at 52 weeks is particularly noteworthy, as weight regain is common following weight loss interventions. Anderson, et al. [34] found that structured programs typically show 50% weight regain by 5 years. Several factors may have contributed to the sustained success observed here. The gradual transition from structured meal replacement to self-selected meals during weeks 8–52 allowed skills acquisition while maintaining caloric control. The preservation of muscle mass observed in our cohort study (+2.33%) [18] may have helped maintain resting metabolic rate, counteracting the metabolic adaptation that commonly undermines long-term weight maintenance. Furthermore, the comprehensive physical activity protocol may have partially mitigated adaptive thermogenesis [33]. The continued weight loss during the maintenance phase, rather than plateau or regain, suggests that participants successfully internalized the healthy behaviors promoted during the intensive phase (Figure 2).
International public health implications
From a global public health perspective, ILM+MR interventions offer several strategic advantages. Unlike bariatric surgery, which requires specialized surgical centers and carries operative risks, lifestyle interventions can be delivered in primary care and community settings, potentially reaching larger populations [35]. This scalability is particularly relevant in ASEAN countries, where several nations have explicitly identified obesity as a priority intervention area within their National Plans of Action for Nutrition [5,6]. The direct and indirect costs of overweight and obesity in Southeast Asia ($11.3 billion in 2018) underscore the urgent need for cost-effective, population-level solutions.
The fundamental components of ILM—dietary modification, physical activity, and behavioral support—can be culturally adapted while maintaining effectiveness. Meal replacement products can be formulated using culturally appropriate ingredients, and behavioral components can be delivered through community-based structures. In countries across the Asia-Pacific region, where traditional diets and eating habits are deeply ingrained, MR-based programs may offer an advantage over conventional dietary counseling by simplifying adherence through portion-controlled, nutritionally complete products [9,28]. The emergence of digital health platforms, including wearable technology, mobile health applications, and telehealth-enabled coaching, offers additional opportunities for scaling ILM programs across the region, where smartphone penetration and digital health adoption are rapidly expanding [36].
Several Southeast Asian countries have launched community-based intervention programs to address the obesity epidemic. Indonesia’s Healthy Living Community Movement (GERMAS), Thailand’s national nutrition programs, and Singapore’s National Steps Challenge represent diverse approaches to promoting healthier lifestyles [5,6]. ILM+MR programs could complement these existing public health initiatives by providing structured, evidence-based weight management within primary care frameworks, particularly for individuals with established obesity who require more intensive intervention than population-level health promotion alone can provide.
Alignment with international guidelines
The findings align with recommendations from major international guideline documents. The 2013 AHA/ACC/TOS guidelines recommend comprehensive lifestyle intervention as first-line treatment for obesity, with specific endorsement of meal replacement strategies [10]. The UK’s NICE guidelines recommend meal replacement products for adults who have not achieved weight loss goals through diet and exercise alone [37]. The Obesity Medicine Association identifies meal replacements as an evidence-based option within medical weight management programs [38]. The recent South and Southeast Asian consensus statement further emphasizes the importance of comprehensive lifestyle interventions as the foundation of obesity management, with bariatric surgery considered for appropriate patients when weight loss targets are not met [9].
Limitations
This case series has several important limitations that must be acknowledged. The 12 participants represent selected cases with exceptional outcomes and do not represent the full range of responses observed in the larger cohort; the mean weight loss of 38.2 kg substantially exceeds the cohort mean of 14.43 kg. The retrospective design introduces potential biases related to data completeness and participant recall. Body composition data (fat mass, muscle mass, visceral fat) were not consistently available for all cases; detailed metabolic assessments including glucose, lipid profiles, and inflammatory markers would strengthen the characterization of health improvements. All participants were from Thailand, potentially limiting generalizability to populations with different genetic backgrounds, dietary patterns, and healthcare systems. Potential confounders such as concurrent stress events or unreported dietary modifications were not systematically assessed. Finally, while 52-week follow-up exceeds the duration of many weight loss studies, longer-term data (5+ years) would be valuable for understanding true maintenance trajectories.
Future research directions
Several priority areas for future investigation emerge from these findings. First, multi-center prospective RCTs comparing ILM+MR to ILM alone and usual care across diverse international sites, including Southeast Asian centers, are critically needed to establish comparative effectiveness with intention-to-treat analysis. Second, predictor analysis identifying baseline demographic, metabolic, and psychobehavioral characteristics associated with exceptional response could enable personalized treatment recommendations; emerging approaches such as gut microbiome profiling and metabolic phenotyping may further refine patient selection [39]. Third, mechanistic studies examining changes in appetite-regulating hormones, gut microbiome composition, and metabolic rate adaptation during and after ILM+MR would illuminate the biological pathways underlying sustained weight loss and inform optimization of MR formulations.
Additionally, prospective body composition assessment using gold-standard methods (DXA or MRI) is needed to rigorously characterize the muscle-preserving effects observed in our cohort study (+2.33%) [18]. Digital health integration—including wearable technology, mobile applications, and telehealth-enabled coaching—offers opportunities to enhance ILM+MR scalability across the Asia-Pacific region, particularly during the maintenance phase [36]. Formal cost-effectiveness analysis comparing ILM+MR with other obesity management strategies would inform healthcare policy in low- and middle-income countries, while implementation science studies examining cultural adaptation, task-shifting to community health workers, and integration into primary care frameworks would support program scaling across diverse ASEAN settings [5]. Finally, extended follow-up beyond 52 weeks (5–10 years) is essential to determine whether the weight losses observed are sustained over clinically relevant time horizons for chronic disease prevention [34,35].
This case series illustrates the substantial and sustained weight loss achievable through intensive lifestyle modification combined with meal replacement, with mean weight loss of 38.2 kg (35.3%) and mean waist circumference reduction of 13.0 inches over 52 weeks. While these outcomes represent exceptional cases rather than typical results, they demonstrate the potential of comprehensive lifestyle intervention to produce transformative health improvements.
The findings strongly support our stated hypothesis. All 12 participants achieved clinically significant weight loss (≥5% of initial body weight), with a mean of 35.3%—substantially exceeding the hypothesized threshold and surpassing benchmarks from major international trials including the DPP (7.0%), Look AHEAD (8.6%), and DiRECT (14.3%) (Table 1; Figure 1). Concurrent waist circumference reductions (mean 13.0 inches) confirmed meaningful improvement in central adiposity, moving all participants below the International Diabetes Federation risk thresholds for Asian populations (Figure 3). Furthermore, the sustained weight loss trajectory through 52 weeks, with continued loss during the maintenance phase rather than the plateau or regain commonly reported in the literature (Figure 2), provides additional support for the hypothesis that the ILM+MR intervention can achieve durable outcomes. However, as these cases were selected for exceptional responses and do not represent the full cohort distribution, the generalizability of these findings to all participants in ILM+MR programs requires confirmation through prospective randomized controlled trials.
The findings complement our published cohort study (n = 702) and align with international research including the DiRECT trial, Look AHEAD study, and systematic meta-analyses supporting meal replacement strategies for weight management. ILM+MR programs offer a scalable, culturally adaptable, and cost-effective approach to addressing the obesity epidemic, with particular relevance for the Asia-Pacific region where the burden of obesity is rapidly increasing and access to specialized surgical interventions remains limited. The multi-component intervention addressing dietary, behavioral, physical activity, and psychosocial factors appears to produce synergistic effects exceeding single-component approaches.
Future research should prioritize prospective randomized trials across diverse international sites, predictor analysis for personalized treatment, mechanistic studies, body composition assessment with gold-standard methods, digital health integration, cost-effectiveness evaluation, and implementation science to translate these promising findings into widespread clinical and public health impact. As the global obesity epidemic continues to expand, comprehensive lifestyle intervention incorporating meal replacement represents a valuable, evidence-based tool for clinicians, healthcare systems, and public health authorities worldwide.
Ethical considerations
This study was reviewed and approved by The Ubon Ratchathani University Ethics Committee for Human Research (approval number: UBU-REC-103/2567). The study followed ethical principles outlined in the Declaration of Helsinki. All participants provided written informed consent for publication of their anonymized data. Confidentiality and anonymity of participant information were ensured throughout the study.
Authors’ contributions
NV conceptualized the study, designed the research methodology, conducted data analysis and interpretation, and drafted the manuscript. SP contributed to study design, data collection, and critical revision of the manuscript. RJ and SH contributed to data collection and participant follow-up. CS provided guidance on study conception and proposal development. All authors reviewed and approved the final manuscript.
The authors acknowledge Dr. Gittipat Thaweepkul, our mentor, for invaluable guidance and support throughout this research. We also acknowledge the ILM+MR research team for their dedication to high-quality data acquisition and precise monitoring. We extend appreciation to the consultants, group-therapy facilitators, and operational staff whose coordinated efforts preserved the integrity of long-term follow-up. We thank all participating families whose sustained engagement enabled the generation of meaningful longitudinal insights.
Funding and conflicts of interest
This study received no external funding. The authors declare no competing interests.
- World Health Organization. Obesity and overweight fact sheet. 2021. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
- McKinsey Global Institute. Overcoming obesity: an initial economic analysis. 2014. Available from: https://www.mckinsey.com/~/media/mckinsey/business%20functions/economic%20studies%20temp/our%20insights/how%20the%20world%20could%20better%20fight%20obesity/mgi_overcoming_obesity_full_report.pdf
- Luppino FS, de Wit LM, Bouvy PF, Stijnen T, Cuijpers P, Penninx BWJH, et al. Overweight, obesity, and depression: a systematic review and meta-analysis of longitudinal studies. Arch Gen Psychiatry. 2010;67:220-229. Available from: https://doi.org/10.1001/archgenpsychiatry.2010.2
- Mottillo S, Filion KB, Genest J, Joseph L, Pilote L, Poirier P, et al. The metabolic syndrome and cardiovascular risk: a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56:1113-1132. Available from: https://doi.org/10.1016/j.jacc.2010.05.034
- Tee ES, Voon SH. Combating obesity in Southeast Asian countries: current status and the way forward. Global Health J. 2024;8(3):147-151. Available from: https://doi.org/10.1016/j.glohj.2024.08.006
- IQVIA. Weighing the crisis: the obesity imperative in Southeast Asia. White paper. 2024. Available from: https://www.iqvia.com/locations/asia-pacific/library/white-papers/weighing-the-crisis-the-obesity-imperative-in-southeast-asia
- Aekplakorn W, Porapakkham Y, Taneepanichskul S. Prevalence of overweight and obesity in Thai population: results of the health examination surveys. J Med Assoc Thai. 2020;103:215-225.
- WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157-163. Available from: https://doi.org/10.1016/s0140-6736(03)15268-3
- Tham KW, Ghani RA, Cua SC, Deerochanawong C, Fojas M, Hocking S, et al. Obesity in South and Southeast Asia—a new consensus on care and management. Obes Rev. 2023;24:e13520. Available from: https://doi.org/10.1111/obr.13520
- Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, Donato KA, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults. Circulation. 2014;129:S102-S138. Available from: https://doi.org/10.1161/01.cir.0000437739.71477.ee
- Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393-403. Available from: https://doi.org/10.1056/nejmoa012512
- Diabetes Prevention Program Research Group. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet. 2009;374:1677-1686. Available from: https://doi.org/10.1016/S0140-6736(09)61457-4
- Look AHEAD Research Group. Eight-year weight losses with an intensive lifestyle intervention: the Look AHEAD study. Obesity. 2014;22:5-13. Available from: https://doi.org/10.1002/oby.20662
- Franz MJ, VanWormer JJ, Crain AL, Boucher JL, Histon T, Caplan W, et al. Weight-loss outcomes: a systematic review and meta-analysis of weight-loss clinical trials with a minimum 1-year follow-up. J Am Diet Assoc. 2007;107:1755-1767. Available from: https://doi.org/10.1016/j.jada.2007.07.017
- Dombrowski SU, Knittle K, Avenell A, Araujo-Soares V, Sniehotta FF. Long term maintenance of weight loss with non-surgical interventions in obese adults: systematic review and meta-analyses of randomised controlled trials. BMJ. 2014;348:g2646. Available from: https://doi.org/10.1136/bmj.g2646
- Heymsfield SB, van Mierlo CAJ, van der Knaap HCM, Heo M, Frier HI. Weight management using a meal replacement strategy: meta and pooling analysis from six studies. Int J Obes. 2003;27:537-549. Available from: https://doi.org/10.1038/sj.ijo.0802258
- Lean MEJ, Leslie WS, Barnes AC, Brosnahan N, Thom G, McCombie L, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. 2018;391:541-551. Available from: https://doi.org/10.1016/s0140-6736(17)33102-1
- Vanichakulthada N, Pitchaiprasert S, Jaroenying R, Hantragool S, Samhugkanee C. Efficacy of intensive lifestyle modification with meal replacement for sustainable weight loss and improved body composition in obese adults: a retrospective cohort study. Open Access J Clin Path Res. 2025;1(2):1-9. Available from: https://doi.org/10.61440/OAJCPR.2025.v1.26
- Wing RR, Tate DF, Gorin AA, Raynor HA, Fava JL. A self-regulation program for maintenance of weight loss. N Engl J Med. 2006;355:1563-1571. Available from: https://doi.org/10.1056/nejmoa061883
- Wing RR, Phelan S. Long-term weight loss maintenance. Am J Clin Nutr. 2005;82:222S-225S. Available from: https://doi.org/10.1093/ajcn/82.1.222s
- Donnelly JE, Blair SN, Jakicic JM, Manore MM, Rankin JW, Smith B, et al. American College of Sports Medicine position stand: appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc. 2009;41:459-471. Available from: https://doi.org/10.1249/mss.0b013e3181949333
- Patel SR, Hu FB. Short sleep duration and weight gain: a systematic review. Obesity. 2008;16:643-653. Available from: https://doi.org/10.1038/oby.2007.118
- Janssen I, Katzmarzyk PT, Ross R. Waist circumference and not body mass index explains obesity-related health risk. Am J Clin Nutr. 2004;79:379-384. Available from: https://doi.org/10.1093/ajcn/79.3.379
- Pischon T, Boeing H, Hoffmann K, Bergmann M, Schulze MB, Overvad K, et al. General and abdominal adiposity and risk of death in Europe. N Engl J Med. 2008;359:2105-2120. Available from: https://doi.org/10.1056/nejmoa0801891
- Nackers LM, Ross KM, Perri MG. The association between rate of initial weight loss and long-term success in obesity treatment: does slow and steady win the race? Int J Behav Med. 2010;17:161-167. Available from: https://doi.org/10.1007/s12529-010-9092-y
- Buchwald H, Estok R, Fahrbach K, Banel D, Jensen MD, Pories WJ, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122:248-256. Available from: https://doi.org/10.1016/j.amjmed.2008.09.041
- Schauer PR, Bhatt DL, Kirwan JP, Wolski K, Brethauer SA, Navaneethan SD, et al. Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med. 2017;376:641-651. Available from: https://doi.org/10.1056/nejmoa1401329
- Ard J, Fitch A, Fruh S, Herman L. Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Adv Ther. 2021;38:2821-2839. Available from: https://doi.org/10.1007/s12325-021-01710-0
- Baumeister RF, Vohs KD, Tice DM. The strength model of self-control. Curr Dir Psychol Sci. 2007;16:351-355. Available from: https://doi.org/10.1111/j.1467-8721.2007.00534.x
- Bandura A. Social foundations of thought and action: a social cognitive theory. Prentice-Hall; 1986. Available from: https://doi.org/10.4135/9781446221129.n6
- Raynor HA, Champagne CM. Position of the Academy of Nutrition and Dietetics: interventions for the treatment of overweight and obesity in adults. J Acad Nutr Diet. 2016;116:129-147. Available from: https://doi.org/10.1016/j.jand.2015.10.031
- Lally P, van Jaarsveld CHM, Potts HWW, Wardle J. How are habits formed: modelling habit formation in the real world. Eur J Soc Psychol. 2010;40:998-1009. Available from: https://psycnet.apa.org/doi/10.1002/ejsp.674
- Rosenbaum M, Hirsch J, Gallagher DA, Leibel RL. Long-term persistence of adaptive thermogenesis in subjects who have maintained a reduced body weight. Am J Clin Nutr. 2008;88:906-912. Available from: https://doi.org/10.1093/ajcn/88.4.906
- Anderson JW, Konz EC, Frederich RC, Wood CL. Long-term weight-loss maintenance: a meta-analysis of US studies. Am J Clin Nutr. 2001;74:579-584. Available from: https://doi.org/10.1093/ajcn/74.5.579
- Loveman E, Frampton GK, Shepherd J, Picot J, Cooper K, Bryant J, et al. The clinical effectiveness and cost-effectiveness of long-term weight management schemes for adults: a systematic review. Health Technol Assess. 2011;15:1-182. Available from: https://doi.org/10.3310/hta15020
- InsightAce Analytic. Digital health for obesity market: global and Asia-Pacific perspectives. 2024.
- National Institute for Health and Care Excellence. Obesity: identification, assessment and management. Clinical guideline. 2014. Available from: https://www.nice.org.uk/guidance/cg189
- Bays HE, McCarthy W, Christensen S. Obesity Algorithm eBook. Obesity Medicine Association; 2021. Available from: https://www.scribd.com/document/558035972/2021-Obesity-Algorithm-1
- Zeevi D, Korem T, Zmora N, Israeli D, Rothschild D, Weinberger A, et al. Personalized nutrition by prediction of glycemic responses. Cell. 2015;163:1079-1094. Available from: https://doi.org/10.1016/j.cell.2015.11.001